光学異性体の非自明性: CAFC Docket No. 2006-1530, -1555
【背景】
King社が販売する降圧薬であるramipril
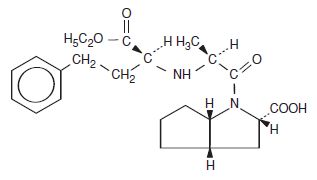
(商標名: Altace; Angiotensin-Converting Enzyme (ACE) inhibitor)について、ジェネリックメーカーであるLupin社がFDAにANDA申請したことに対し、ramiprilをカバーするUS 5,061,722特許を侵害しているとして特許権者であるAventis社及びそのライセンシーであるKing社が訴訟を提起した。
ramiprilはその有する不斉炭素原子5つ全てが”S”の立体配置(”5(S)”)となっている、2の5乗つまり32の立体異性体ファミリーのうちのひとつである。
Lupin社は、5(S)体及びSSSSR体の混合物(SCH 31925 mixture)を記載した文献及び、異性体である類薬enalapril
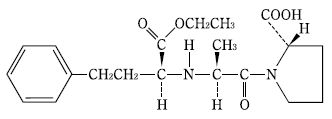
に関する文献等から自明であること等を理由に、該特許は無効であると主張しが、地裁は特許有効であると判断、Lupin社は控訴した。
Claims:
1. A compound of the formula
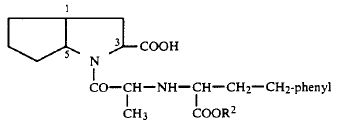
or a physiologically acceptable salt thereof, wherein R2 is hydrogen, methyl, ethyl, or benzyl, and wherein hydrogen atoms on the ring carbon atoms in the 1- and 5-positions are in the cis-configuration relative to one another, the carboxyl group on the ring carbon atom in the 3-position is in the endo position relative to the bicyclic ring system, and the chirality centers in the chain and on the ring carbon atom in the 3-position all have the S-configuration, said compound or salt being substantially free of other isomers.
2. A compound or salt as in claim 1 which is N-(1-S-carboethoxy-3-phenyl-propyl)-S-alanyl-cis,endo-2-azabicyclo-[3.3.0]-octane-3-S-carboxylic acid or a salt thereof.
(クレーム1の化合物は、R2がethylのとき、ramiprilであり、クレーム2はramiprilをspecificにカバーしている。他のクレームは省略。)
【要旨】
CAFCは、KSR事件(自明性判断に関する最高裁判決)に触れ、下記のように自明性判断の一般的考え方について言及した。
It remains necessary to show “‘some articulated reasoning with some rational underpinning to support the legal conclusion of obviousness,’” but such reasoning “need not seek out precise teachings directed to the specific subject matter of the challenged claim.” See id. (quoting In re Kahn, 441 F.3d 977, 988 (Fed. Cir. 2006)). Requiring an explicit teaching to purify the 5(S) stereoisomer from a mixture in which it is the active ingredient is precisely the sort of rigid application of the TSM test that was criticized in KSR.
The “reason or motivation” need not be an explicit teaching that the claimed compound will have a particular utility; it is sufficient to show that the claimed and prior art compounds possess a “sufficiently close relationship . . . to create an expectation,” in light of the totality of the prior art, that the new compound will have “similar properties” to the old.
Once such a prima facie case is established, it falls to the applicant or patentee to rebut it, for example with a showing that the claimed compound has unexpected properties.
if it is known that some desirable property of a mixture derives in whole or in part from a particular one of its components, or if the prior art would provide a person of ordinary skill in the art with reason to believe that this is so, the purified compound is prima facie obvious over the mixture even without an explicit teaching that the ingredient should be concentrated or purified.
Ordinarily, one expects a concentrated or purified ingredient to retain the same properties it exhibited in a mixture, and for those properties to be amplified when the ingredient is concentrated or purified; isolation of interesting compounds is a mainstay of the chemist’s art. If it is known how to perform such an isolation, doing so “is likely the product not of innovation but of ordinary skill and common sense.” KSR, 127 S. Ct. at 1742.
上記自明性判断の考え方に基づき、CAFCは本事案を下記のように当てはめ判断した。
The SCH 31925 composition contained only the 5(S) and SSSSR stereoisomers of ramipril. Importantly, these forms differ by the configuration of only one carbon atom, and that atom is not one of the “bridgehead” carbons. Rather, that carbon atom is in the part of the ramipril molecule that is common to the enalapril molecule. In enalapril, as in captopril and BPP5a before it, all of the stereocenters are in the S configuration; the Merck article taught that the SSS configuration of enalapril is 700 times as potent as the SSR form. The close structural analogy between 5(S) and SSSSR ramipril and SSS and SSR enalapril would have led a person of ordinary skill to expect 5(S) and SSSSR ramipril to differ similarly in potency. Moreover, the ’944 patent specifically taught that stereoisomers of ramipril “can be separated by conventional chromatographic or fractional crystallization methods.” ’944 patent, col. 10, ll. 28–31. Aventis’s protestations notwithstanding, there is no evidence that separating 5(S) and SSSSR ramipril was outside the capability of an ordinarily skilled artisan.
The prior art supporting prima facie obviousness included the SCH 31925 mixture, and so Aventis must show that 5(S) ramipril had unexpected results not over all of its stereoisomers, but over that mixture, which did not contain the RRSSS form.
‘712特許は自明。侵害と判断した地裁判断を覆した。
【コメント】
光学異性体の非自明性が争われたケース。
特許権者は、異性体の分離困難性及びunexpected resultsを示すことができず、prima facie obviousnessを反証することができなかった。この反証材料があるかどうかが光学異性体の非自明性を認めうる決め手のようである。
上記判決は、KSR事件(自明性判断に関する最高裁判決)後における米国での光学異性体の自明性判断のボーダーラインを検討するために参考になると考えられる。

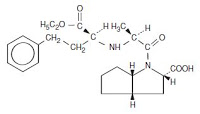


コメント